Objet Ltd., an innovation leader in 3D printing for rapid prototyping and additive manufacturing, demonstrated the benefits of adding 3D printing to the dental workflow at the International Expodental Rome.
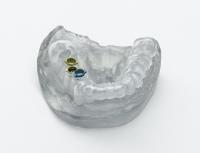 Objet presented the new Bio-Compatible 3D printing material (MED610™) and its 3D printers that help dental labs to digitize their dentistry for more accurate and cost effective results. According to Objet, the material combines bio-compatibility with high dimensional stability and clear transparency. This makes it useful for PMMA simulation and a range of dental solutions – particularly the production of accurate, custom surgical guides.
Objet presented the new Bio-Compatible 3D printing material (MED610™) and its 3D printers that help dental labs to digitize their dentistry for more accurate and cost effective results. According to Objet, the material combines bio-compatibility with high dimensional stability and clear transparency. This makes it useful for PMMA simulation and a range of dental solutions – particularly the production of accurate, custom surgical guides.
Avi Cohen, Head of Medical Solutions at Objet added, “The advanced mechanical properties of the new Bio-Compatible material, including clear transparency, bring benefits to the entire medical and dental workflow – from surgical planning through to the procedure itself.”
 The new material is suitable for prolonged skin contact of more than 30 days and mucosal-membrane contact of up to 24 hours. It has five medical approvals according to the harmonized standard ISO 10993‑1: Cytotoxicity, Genotoxicity, Delayed Type Hypersensitivity, Irritation and USP Plastic Class VI.* MED610 is also manufactured under the ISO 13485:2003 certification, which specifies that each and every batch of the material undergoes bio-compatibility conformity testing, including GC-FID before it is packaged. This ensures the highest bio-compatible standards for dental solutions requirements.
The new material is suitable for prolonged skin contact of more than 30 days and mucosal-membrane contact of up to 24 hours. It has five medical approvals according to the harmonized standard ISO 10993‑1: Cytotoxicity, Genotoxicity, Delayed Type Hypersensitivity, Irritation and USP Plastic Class VI.* MED610 is also manufactured under the ISO 13485:2003 certification, which specifies that each and every batch of the material undergoes bio-compatibility conformity testing, including GC-FID before it is packaged. This ensures the highest bio-compatible standards for dental solutions requirements.
Objet also showcased a range of the dental models at the event, specifically designed for the dental market. The material features extreme toughness, dimensional stability and great detail visualization – producing models that can be handled immediately after being built.
Among dental laboratories already using Objet 3D printing technology are Glidewell Laboratories, Albensi Laboratories, iDent Imaging and Remedent to fabricate unique stone models, orthodontic appliances, delivery and positioning trays, veneer try-ins, full and partial denture try-ins, surgical guides, clear aligners and retainers.
Objet Ltd.
www.ObjetDental.com
*Biological Testing: Parts printed by Objet according to Objet MED610 Use and Maintenance Terms (DOC-08242) were evaluated for biocompatibility in accordance with standard DIN EN ISO 10993-1: 2009, Biological evaluation of medical devices-Part 1: Evaluation and Testing within a risk management process. This addresses cytotoxicity, genotoxicity, delayed hypersensitivity, and USP plastic Class VI which includes the test for irritation, acute systemic toxicity and implantation.


Leave a Reply
You must be logged in to post a comment.